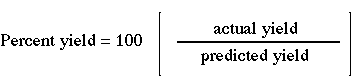Percent Yield
Many reactions fail to give a 100% yield of product. Main reasons:
The theoretical yield of a reactions is the calculated amount of product that should be obtained. In order to determine this amount, we must have a balanced chemical equation and do a stoichiometry problem.
The actual yield of a reaction is the amount of product that forms when we got to the lab and perform the experiment. This amount cannot be calculated but must be measured in lab or given in the problem.
The percent yield is the ratio of the actual yield to the theoretical yield multiplied by 100. Both the theoretical and the actual yields must have the same units to obtain a percent. Calculating percent yield measures the efficiency of the reaction in changing the reactants to products
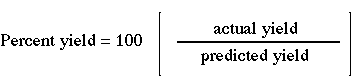
- Reactions do not always go to completion or may be reversible.
- Impure reactants and competing side reactions may cause other products to form.
- Some product may be lost in handling and transferring from one reaction vessel to another.
The theoretical yield of a reactions is the calculated amount of product that should be obtained. In order to determine this amount, we must have a balanced chemical equation and do a stoichiometry problem.
The actual yield of a reaction is the amount of product that forms when we got to the lab and perform the experiment. This amount cannot be calculated but must be measured in lab or given in the problem.
The percent yield is the ratio of the actual yield to the theoretical yield multiplied by 100. Both the theoretical and the actual yields must have the same units to obtain a percent. Calculating percent yield measures the efficiency of the reaction in changing the reactants to products